An Introduction to Transfection
Transfection refers to the deliberate introduction of nucleic acids—such as DNA, RNA, or oligonucleotides—into eukaryotic cells through nonviral delivery methods. This molecular biology technique is fundamental for manipulating gene expression in vitro, enabling researchers to study the functional consequences of gene activation, repression, or mutation within controlled cellular environments. It is routinely employed in gene function analysis, protein expression studies, RNA interference experiments, and therapeutic gene delivery development.
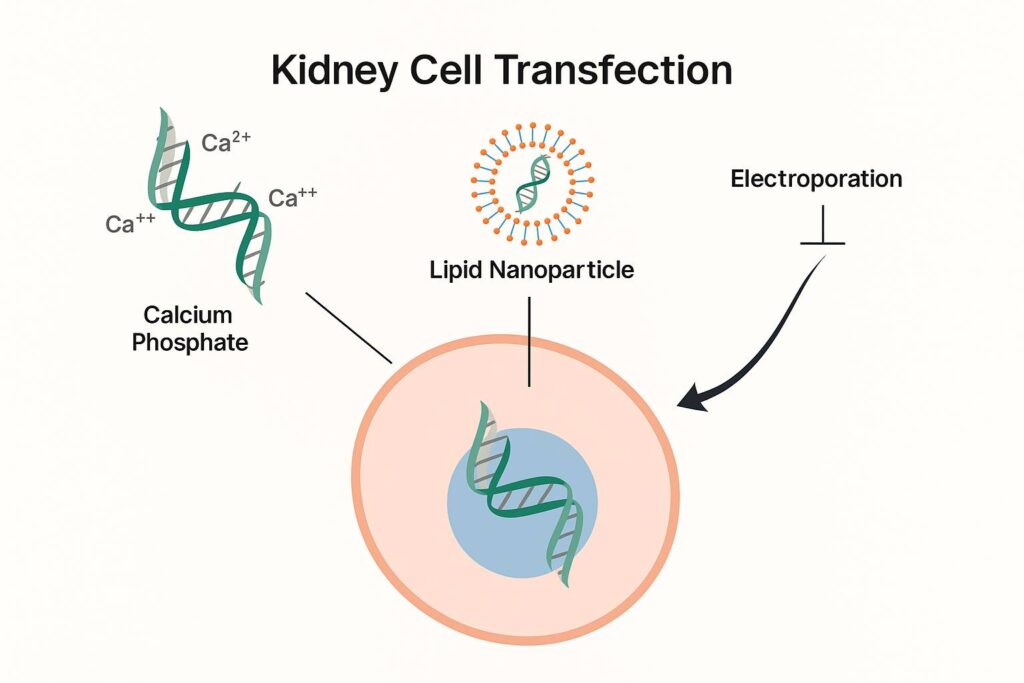
The process of transfection addresses the biophysical challenge of delivering highly anionic nucleic acids across the negatively charged phospholipid bilayer of the cell membrane. To overcome this electrostatic barrier, various chemical and physical methods have been developed. Chemical strategies include the use of calcium phosphate precipitates, polycationic compounds such as diethylaminoethyl (DEAE)-dextran, and synthetic cationic lipids. These reagents form electrostatic complexes with nucleic acids, thereby neutralizing or reversing their net charge and facilitating cellular uptake via endocytosis or membrane fusion. Lipid-based vectors, particularly those containing fusogenic components, promote integration of the complex into the lipid bilayer, enhancing cytoplasmic delivery.
Physical methods, such as microinjection and electroporation, offer direct intracellular delivery by transiently disrupting the plasma membrane. Electroporation, for example, uses short high-voltage electrical pulses to induce membrane permeabilization, allowing nucleic acids to diffuse into the cytosol. Microinjection bypasses the membrane entirely by mechanically inserting nucleic acids into the cytoplasm or nucleus using fine-tipped glass needles.
The advent of reporter gene constructs and selection markers has expanded the analytical applications of transfection. These tools enable both transient expression and stable genomic integration of exogenous sequences, thereby supporting investigations into transcriptional regulation, post-transcriptional processing, translational control, and protein-protein or protein-DNA interactions. Transfection continues to be a cornerstone of molecular and cellular biology, underpinning advances in basic research, drug discovery, and the development of gene-based therapeutics.
An Introduction to Kidney Transfection
Kidney transfection refers to the delivery of nucleic acids, including plasmid DNA, small interfering RNA (siRNA), messenger RNA (mRNA), or antisense oligonucleotides, into renal cells in order to manipulate gene expression for experimental or therapeutic purposes. This molecular technique is central to renal research, offering a means to investigate gene function, explore pathophysiological mechanisms of kidney disease, and develop gene-based therapeutic strategies targeting nephron compartments, renal epithelium, or glomerular structures.
The kidney presents several anatomical and physiological barriers that complicate efficient nucleic acid delivery. Renal epithelial cells exhibit strong apical-basal polarity, are tightly connected via junctional complexes, and often reside within dense extracellular matrices, all of which hinder nucleic acid uptake. Additionally, systemic administration of nucleic acids frequently results in hepatic clearance or degradation before reaching renal tissue. Therefore, effective kidney transfection requires cell-type-specific optimization of both delivery methods and vector systems, particularly when working with differentiated renal cell types such as podocytes, proximal tubule epithelial cells, and collecting duct cells.
In vitro transfection of kidney-derived cell lines, including HEK293 and HK-2 cells, is typically achieved using lipid-based reagents, cationic polymers like polyethylenimine (PEI), or electroporation techniques. These methods enable either transient or stable transgene expression and are used to model renal ion transport, cellular signaling pathways, and injury responses in culture systems. In vivo transfection of kidney tissue, however, requires more sophisticated approaches to ensure sufficient transgene uptake and specificity. Hydrodynamic injection, often administered via the tail vein or renal vein, creates transient high-pressure conditions that facilitate the entry of nucleic acids into renal parenchymal cells. Other techniques, such as ultrasound-targeted microbubble destruction (UTMD), direct renal electroporation, or nanoparticle-assisted delivery, have been developed to enhance uptake and reduce systemic distribution.
Recent innovations in nanoparticle design have further improved the selectivity and efficiency of renal gene delivery. Targeted nanoparticles, incorporating ligands that bind renal-specific receptors such as megalin, cubilin, or folate receptors, have shown increased localization and uptake in kidney tissues while minimizing off-target expression. Additionally, advances in organoid modeling and ex vivo perfusion systems now allow for more refined and physiologically relevant transfection experiments using human-derived kidney tissue.
Kidney transfection has become an essential tool in nephrology and molecular medicine, enabling researchers to study the roles of renal transporters, receptors, and transcription factors in both normal physiology and disease. It supports gene silencing approaches for therapeutic target validation in models of nephropathy and fibrosis, facilitates CRISPR/Cas9-mediated gene editing in renal cells and organoids, and allows for mRNA-based delivery strategies aimed at promoting regenerative responses or modulating inflammation. Transfection also enables high-throughput screening of oligonucleotide-based drugs in acute kidney injury (AKI) and chronic kidney disease (CKD) models. As delivery platforms continue to improve, kidney transfection remains at the forefront of renal research and therapeutic innovation.
When a gene is transferred into the kidney, it might be expressed in the tubular or glomerular regions. To specifically target designated kidney cells for therapeutic reasons, it is advantageous to be able to regulate the transgene’s final destination. Transfection of proximal tubular cells by polyplex infusion into the rat renal artery was recently shown. Fluorescent polyethyleneimine polyplexes with fluorescent poly-L-lysine were created to explore if this mechanism involved glomerular filtration of the DNA-carrying particles. This way, it was possible to see precisely how the particles made their way into the kidney. Since luminous proximal tubules were found in our polyplexes, they were filtered via the glomerulus. DOTAP-containing luminous lipo-polyplexes, on the other hand, were never seen in tubular cells. It was found the mean size of polyplexes was less than lipo0polyplexes (93 nm) via laser light scattering (160 nm). Because of the restrictions given by the glomerular filtration membrane, the diameter of the transfecting particles is an essential element in this process. This knowledge is significant in the context of optimizing gene transcription in tubular cells by manipulating the physical and chemical characteristics of DNA complexes.
Innovative Non-Viral Gene Delivery for Renal Tissue
At the core of our transfection technology is a proprietary nanoparticle-liposome complex designed for enhanced delivery of nucleic acids to kidney cells and tissues. This advanced system offers a non-viral, biocompatible alternative to traditional gene transfer methods, ensuring high transfection efficiency while minimizing toxicity, inflammation, and off-target effects.
Kidney tissues present unique challenges for gene delivery due to complex vascular structures, active filtration barriers, and heterogeneous cell populations. Our technology overcomes these barriers by leveraging charge-optimized liposomal carriers that form stable complexes with siRNA, miRNA, mRNA, or plasmid DNA. These complexes facilitate efficient cellular uptake, endosomal escape, and functional expression or knockdown of target genes.
Mechanism of Action
Once administered via systemic (IV, IP) or localized (intrarenal, subcapsular, or tumor-directed) injection, the transfection complex circulates in serum while remaining stable and bioavailable. Targeted uptake occurs predominantly in renal cortex and tumor tissue, where nucleic acids are released intracellularly to elicit gene silencing or expression. The platform supports both transient transfection and co-delivery protocols, enabling simultaneous introduction of multiple constructs (e.g., siRNA with plasmid DNA).
Applications in Renal Research
This delivery system has been validated in a variety of experimental contexts, including:
- Functional genomics studies in renal cell carcinoma and chronic kidney disease models
- Evaluation of nephroprotective gene targets using in vivo RNA interference
- Gene expression profiling of drug transporters and fibrotic markers in toxicology studies
- Co-transfection studies for modeling pathway interactions and epigenetic modulation
Advantages of Our Kidney Transfection Technology
- Serum stability: Maintains activity for up to 24 hours post-injection
- Biodegradable formulation: Minimizes immune response and systemic toxicity
- Kidney specificity: Accumulates preferentially in renal tissues and tumors
- Validated performance: Confirmed by qRT-PCR, western blotting, and in vivo imaging
Our technology is the result of years of development in collaboration with academic laboratories and biotechnology firms. By integrating this delivery platform with Altogen Labs’ in vivo research services, we offer complete gene modulation solutions that meet the highest standards in reproducibility, safety, and scientific precision.
1. 786‑O Xenograft Model
A clear‑cell renal cell carcinoma (RCC) model derived from a 58-year-old male patient. This hypertriploid cell line expresses VEGF and PTH, supporting tumor angiogenesis and growth. It’s frequently used for evaluating anti-angiogenic therapies and COX‑2 inhibitors, showing improved efficacy when drugs like celecoxib are combined with first-line agents like sunitinib
Learn more about the 786‑O model
2. A498 Xenograft Model
Derived from the renal carcinoma of a 52-year-old male in the 1970s, this epithelial-rich cell line is commonly used in preclinical RCC studies. It lacks functional VHL and expresses HIF‑2α constitutively. Recent work has demonstrated its utility in evaluating combined therapies targeting angiogenesis (lenvatinib + everolimus) and HIF pathways (e.g., YC‑1)
Learn more about the A498 model
3. Caki‑1 Xenograft Model
This metastatic clear-cell RCC cell line originates from a skin metastasis of a 49-year-old patient. It features polarized epithelial characteristics and is widely used to assess anti-angiogenic (sunitinib, sorafenib) and RAF-1/PDGFR inhibitors, as well as combination chemotherapy (5-FU) in vivo
Learn more about the Caki‑1 model
4. G401 Xenograft Model
A pediatric kidney rhabdoid tumor line, derived from a 3-month-old male in 1973. G401 is a robust model for pediatric renal carcinomas and has been instrumental in testing oncolytic viruses (e.g., M002) and epigenetic therapies (HDAC and EZH2 inhibitors), demonstrating differentiation and tumor suppression in vivo
Learn more about the G401 model
5. Renca Syngeneic Murine Model
A murine Balb/c-derived renal carcinoma model widely used for immune-oncology research due to its syngeneic nature. It closely mimics clear cell RCC, exhibits spontaneous metastasis, and is ideal for studying tumor-immune interactions in immunocompetent hosts
