786-O Kidney Cancer Xenograft Model
Altogen Labs provides specialized preclinical research services utilizing the 786-O renal cell carcinoma (RCC) xenograft model. The 786-O cell line, derived from a primary clear cell adenocarcinoma, is characterized by the absence of functional VHL and HIF-1α expression, coupled with high constitutive HIF-2α levels, making it a pertinent model for studying RCC pathophysiology.
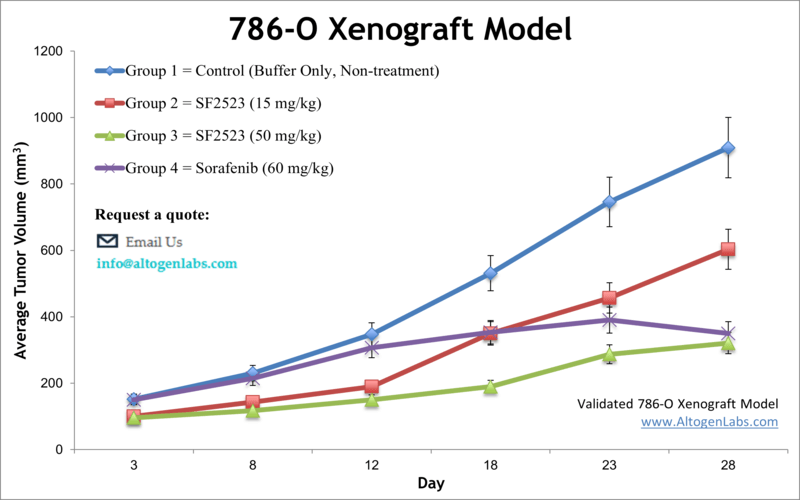
Key Features of the 786-O Xenograft Model:
- Tumor Establishment: Subcutaneous implantation into immunocompromised mice (e.g., NOD/SCID or athymic nude strains) with tumor growth typically observable within 4–7 days post-implantation.
- Treatment Administration: Compounds are administered according to customized dosing schedules once tumors reach a predetermined size (typically 50–100 mm³).
- Monitoring and Data Collection: Tumor volumes are measured regularly, and mouse weights are recorded multiple times weekly to assess treatment efficacy and tolerability.
- Study Endpoints: Animals are euthanized when tumors reach a size limit (e.g., 2,000 mm³) or as defined by the study protocol, followed by necropsy and tissue collection for downstream analyses.
Available Analyses and Services:
- Efficacy Assessments: Tumor Growth Delay (TGD) and Tumor Growth Inhibition (TGI) studies to evaluate therapeutic impact.
- Histological and Molecular Analyses: Immunohistochemistry, gene expression profiling, and protein/RNA isolation from tumor tissues.
- Safety and Toxicology: Blood chemistry analysis and comprehensive toxicity evaluations.
- Additional Services: Lipid distribution studies, metabolic assays, and imaging studies to support comprehensive pharmacological profiling.
Request an Instant Quote: https://altogenlabs.com/request-quote/786o-xenograft-model-services/
Link to Service: 786-O Xenograft Model
A498 Kidney Cancer Xenograft Model
Altogen Labs offers comprehensive preclinical research services utilizing the A498 renal cell carcinoma (RCC) xenograft model, a well-characterized system for studying clear cell RCC (ccRCC). The A498 cell line, derived from human kidney carcinoma, exhibits epithelial-like adherent properties and a hypertriploid karyotype. It expresses vimentin and cytokeratins 8 and 18, while lacking expression of cytokeratins 7, 17, 19, desmin, EpCAM, and neurofilament, making it a pertinent model for investigating RCC pathophysiology and therapeutic responses.
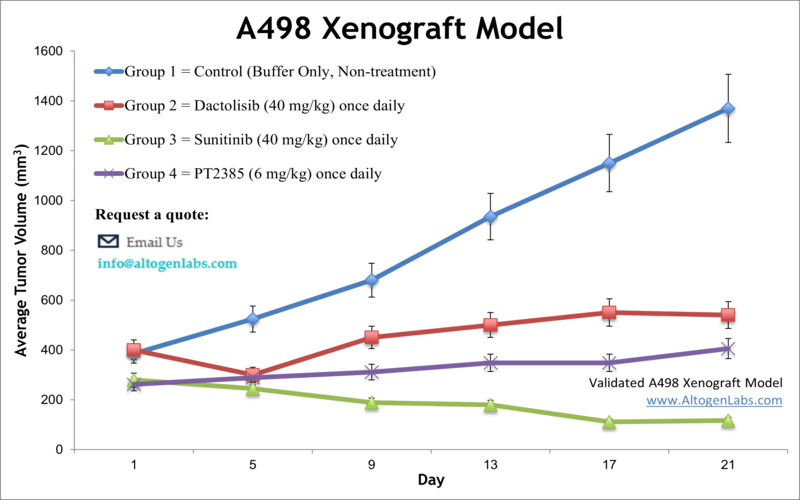
Key Features of the A498 Xenograft Model
- Tumor Establishment: Subcutaneous implantation of A498 cells into immunocompromised mice (e.g., NOD/SCID or athymic nude strains) leads to tumor formation, enabling in vivo assessment of tumor growth and response to therapies.
- Treatment Administration: Compounds are administered according to customized dosing schedules once tumors reach a predetermined size (typically 80–120 mm³).
- Monitoring and Data Collection: Tumor volumes are measured regularly, and mouse weights are recorded multiple times weekly to assess treatment efficacy and tolerability.
- Study Endpoints: Animals are euthanized when tumors reach a size limit (e.g., 2,000 mm³) or as defined by the study protocol, followed by necropsy and tissue collection for downstream analyses.
Available Analyses and Services
- Efficacy Assessments:
- Tumor Growth Delay (TGD): Evaluation of latency in tumor development.
- Tumor Growth Inhibition (TGI): Quantitative measurement of tumor volume reduction in response to therapeutic agents.
- Histological and Molecular Analyses:
- Immunohistochemistry (IHC): Evaluation of biomarkers to assess treatment effects.
- Gene Expression Profiling: Analysis of treatment-induced changes in gene expression using qRT-PCR and Western blotting.
- Safety and Toxicology:
- Blood Chemistry Analysis: Monitoring of systemic toxicity and organ function.
- Histopathological Examination: Detailed tissue analysis to identify potential adverse effects.
- Advanced Therapeutic Studies:
- RNA Interference (RNAi): In vivo delivery of siRNA/shRNA to target specific genes involved in tumor progression.
- Liposome Encapsulation: Formulation of therapeutic agents for enhanced delivery and efficacy.
- Imaging Studies:
- Fluorescence-based Whole-body Imaging: Visualization of tumor growth and metastasis.
- Magnetic Resonance Imaging (MRI): High-resolution imaging for detailed tumor assessment.
Request an Instant Quote: https://altogenlabs.com/xenograft-models/kidney-cancer-xenograft/a498-xenograft-model/
Link to Service: A498 Xenograft Model
Caki-1 Kidney Cancer Xenograft Model
Altogen Labs offers comprehensive preclinical research services utilizing the Caki-1 xenograft model, a well-established system for studying clear cell renal cell carcinoma (ccRCC). The Caki-1 cell line, derived from a skin metastasis of ccRCC, exhibits characteristics of proximal tubule epithelium and is instrumental in evaluating therapeutic agents targeting angiogenesis and tumor proliferation.
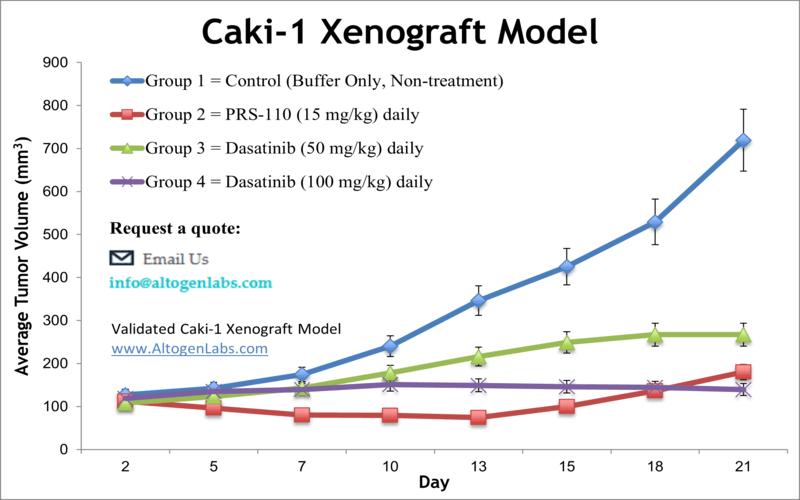
Key Features of the Caki-1 Xenograft Model
- Tumor Establishment: Subcutaneous implantation of Caki-1 cells into immunocompromised mice (e.g., NOD/SCID or athymic nude strains) leads to tumor formation, enabling in vivo assessment of tumor growth and response to therapies.
- Relevance to ccRCC: The model closely mimics human ccRCC, characterized by extensive vasculature and resistance to conventional therapies, making it suitable for evaluating anti-angiogenic agents and combination treatments.
- Therapeutic Evaluation: Caki-1 xenografts have been employed in preclinical studies to assess the efficacy of various therapeutic agents, including combinations of 5-fluorouracil with sorafenib or sunitinib, demonstrating their utility in translational cancer research.
Available Services and Analyses
- Efficacy Studies:
- Tumor Growth Inhibition (TGI): Quantitative measurement of tumor volume reduction in response to therapeutic agents.
- Survival Analysis: Assessment of treatment impact on overall survival in xenograft-bearing mice.
- Molecular and Histological Analyses:
- Immunohistochemistry (IHC): Evaluation of biomarkers to assess treatment effects.
- Gene Expression Profiling: Analysis of treatment-induced changes in gene expression using qRT-PCR and Western blotting.
- Pharmacokinetics and Toxicology:
- Blood Chemistry Analysis: Monitoring of systemic toxicity and organ function.
- Histopathological Examination: Detailed tissue analysis to identify potential adverse effects.
- Advanced Therapeutic Studies:
- RNA Interference (RNAi): In vivo delivery of siRNA/shRNA to target specific genes involved in tumor progression.
- Liposome Encapsulation: Formulation of therapeutic agents for enhanced delivery and efficacy.
Request an Instant Quote: https://altogenlabs.com/request-quote/caki-1-xenograft-model-services/
Link to Service: Caki-1 Xenograft Model
G401 Kidney Cancer Xenograft Model
Altogen Labs offers comprehensive preclinical research services utilizing the G401 kidney cancer xenograft model, a critical tool for studying pediatric renal tumors, particularly malignant rhabdoid tumors (MRTs). The G401 cell line, established from a 3-month-old male patient with a renal rhabdoid tumor, serves as a valuable model for evaluating novel therapeutic strategies against aggressive pediatric kidney cancers.
Key Features of the G401 Xenograft Model
- Tumor Establishment: Subcutaneous implantation of G401 cells into immunocompromised mice (e.g., NOD/SCID or athymic nude strains) leads to tumor formation, enabling in vivo assessment of tumor growth and response to therapies.
- Relevance to Pediatric Oncology: G401 xenografts closely mimic the biological behavior of MRTs, characterized by the inactivation or deletion of the SMARCB1 gene, making them suitable for studying epigenetic therapies and differentiation-inducing agents.
- Therapeutic Evaluation: The model has been employed in preclinical studies to assess the efficacy of various therapeutic agents, including histone deacetylase inhibitors (e.g., panobinostat) and EZH2 inhibitors (e.g., tazemetostat), demonstrating its utility in translational cancer research.
Available Services and Analyses
- Efficacy Studies:
- Tumor Growth Inhibition (TGI): Quantitative measurement of tumor volume reduction in response to therapeutic agents.
- Survival Analysis: Assessment of treatment impact on overall survival in xenograft-bearing mice.
- Molecular and Histological Analyses:
- Immunohistochemistry (IHC): Evaluation of biomarkers such as TUJ1 and PAX2 to assess neuronal and renal differentiation.
- Gene Expression Profiling: Analysis of treatment-induced changes in gene expression using qRT-PCR and Western blotting.
- Pharmacokinetics and Toxicology:
- Blood Chemistry Analysis: Monitoring of systemic toxicity and organ function.
- Histopathological Examination: Detailed tissue analysis to identify potential adverse effects.
- Advanced Therapeutic Studies:
- RNA Interference (RNAi): In vivo delivery of siRNA/shRNA to target specific genes involved in tumor progression.
- Liposome Encapsulation: Formulation of therapeutic agents for enhanced delivery and efficacy.
Request an Instant Quote: Contact us at info@altogenlabs.com
Link to Service: G401 Xenograft Model
Renca Syngeneic Murine Kidney Cancer Model
Altogen Labs offers comprehensive preclinical research services utilizing the Renca syngeneic murine model, an established system for studying renal cell carcinoma (RCC) within an immunocompetent host. Derived from a spontaneous renal adenocarcinoma in BALB/c mice, the Renca cell line closely mimics human clear cell RCC and is particularly valuable for evaluating immunotherapies and metastatic progression.
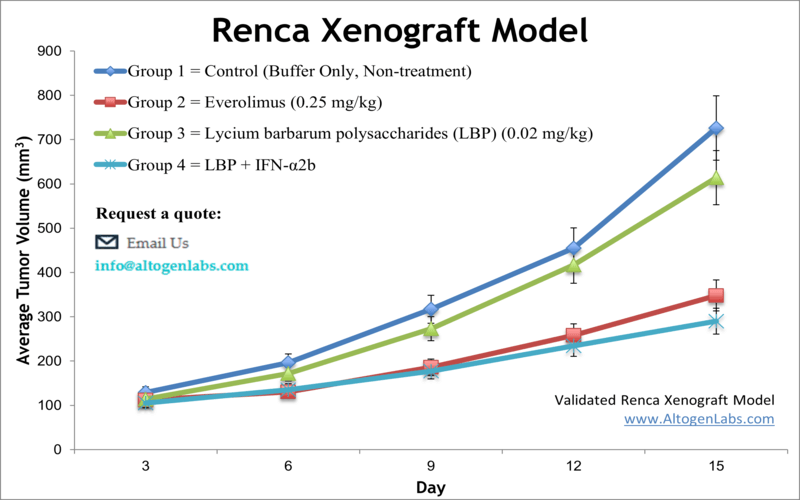
Key Features of the Renca Model
- Immunocompetent Host System: Utilizes BALB/c mice, preserving native immune responses, making it ideal for immuno-oncology studies.
- Metastatic Potential: The Renca model exhibits spontaneous metastasis, allowing for the assessment of anti-metastatic therapies.
- Flexible Tumor Implantation:
- Subcutaneous: For straightforward tumor growth monitoring.
- Orthotopic: Implantation into the kidney to replicate the tumor’s natural environment.
- Intravenous and Intracardiac: For systemic metastasis studies.
Available Services and Analyses
- Efficacy Studies:
- Tumor Growth Delay (TGD): Assessment of latency in tumor development.
- Tumor Growth Inhibition (TGI): Evaluation of therapeutic impact on tumor progression.
- Dosing Regimens:
- Customizable administration routes including intravenous, intraperitoneal, oral gavage, and more, tailored to study requirements.
- Immunohistochemistry (IHC):
- Analysis of tumor tissues for specific biomarkers to elucidate treatment effects.
- Molecular Analyses:
- Isolation of RNA and proteins for gene expression studies.
- Quantitative PCR and Western blotting to assess molecular changes post-treatment.
- Toxicology and Safety Assessments:
- Blood chemistry analysis to monitor systemic effects.
- Comprehensive histopathological evaluations to detect potential toxicities.
- Metabolic and Distribution Studies:
- Lipid distribution and metabolic assays to understand pharmacokinetics and pharmacodynamics.
Request an Instant Quote: https://altogenlabs.com/request-quote/renca-xenograft-model-services/
Link to Service: Renca Xenograft Model
RXF393 Kidney Cancer Xenograft Services by Altogen Labs
Altogen Labs offers comprehensive preclinical research services utilizing the RXF393 renal cell carcinoma (RCC) xenograft model, a valuable system for studying clear cell RCC (ccRCC). The RXF393 cell line, derived from a 53-year-old male patient with renal cancer, is characterized by a TP53 mutation (Arg175), making it pertinent for evaluating therapeutic agents targeting p53-deficient tumors.
Key Features of the RXF393 Xenograft Model
- Tumor Establishment: Subcutaneous implantation of RXF393 cells into immunocompromised mice (e.g., NOD/SCID or athymic nude strains) leads to tumor formation, enabling in vivo assessment of tumor growth and response to therapies.
- Relevance to ccRCC: The model closely mimics human ccRCC, characterized by resistance to conventional therapies, making it suitable for evaluating novel therapeutic agents and combination treatments.
- Therapeutic Evaluation: RXF393 xenografts have been employed in preclinical studies to assess the efficacy of various therapeutic agents, including small molecule inhibitors and immunotherapies, demonstrating their utility in translational cancer research.
Available Services and Analyses
- Efficacy Studies:
- Tumor Growth Inhibition (TGI): Quantitative measurement of tumor volume reduction in response to therapeutic agents.
- Survival Analysis: Assessment of treatment impact on overall survival in xenograft-bearing mice.
- Molecular and Histological Analyses:
- Immunohistochemistry (IHC): Evaluation of biomarkers to assess treatment effects.
- Gene Expression Profiling: Analysis of treatment-induced changes in gene expression using qRT-PCR and Western blotting.
- Pharmacokinetics and Toxicology:
- Blood Chemistry Analysis: Monitoring of systemic toxicity and organ function.
- Histopathological Examination: Detailed tissue analysis to identify potential adverse effects.
- Advanced Therapeutic Studies:
- RNA Interference (RNAi): In vivo delivery of siRNA/shRNA to target specific genes involved in tumor progression.
- Liposome Encapsulation: Formulation of therapeutic agents for enhanced delivery and efficacy.
Request an Instant Quote: Contact us at info@altogenlabs.com
Link to Service: RXF393 Xenograft Model
Compliance and Quality Assurance
All studies are conducted in compliance with Institutional Animal Care and Use Committee (IACUC) regulations and adhere to Good Laboratory Practice (GLP) standards, ensuring ethical treatment of animals and high-quality, reproducible data.
For more detailed information or to request a quote for RXF393 xenograft services, please contact Altogen Labs at info@altogenlabs.com or call (512) 433-6177.
